AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Electron geometry chart diatomic3/19/2024 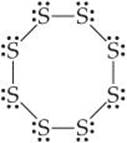
We need to determine the number of valence electrons that this particular an ion possesses Flooring is in group seven a. Right? The electron configuration for the dye forming an ion F to minus one. All right, so keep this in mind as we start doing more and more questions dealing with homo nuclear di atomic molecules Hide transcripts So just keep in mind these are the different types of molecular orbital is that can exist based on what type of period element you're dealing with 12 or three. So we're starting out with two S again and we just start pulling together our atomic orbital electrons and dumping them into our molecular orbital's and start filling it up as we move up. 
So here again, oxygen to neon there's still period two elements. So now my pie toupee moves up And my Sigma two p. If we look the change happens here they flip. We're gonna have a slight change when it comes to die atomic oxygen to die atomic neon. We always fill from lowest molecular orbital up now again because of increasing electro negativity and a decrease in atomic size. Another anti body molecular orbital again. Gets more complex as the atomic orbital's continue to pull their electrons together to create new molecular orbital's here. We fill in first are bonding molecular orbital which is signaled to us and then start filling in our sigma star to us, which represents our anti body molecular orbital. Now here with the two s orbital is atomic orbital's. So we start off with two S and we move our way up to two P. Now if we move to the next one, this correlates to die atomic lithium all the way to die atomic nitrogen here it gets more complex because now we're dealing with period two elements. Represents our anti bonding molecular orbital. Represents our bonding molecular orbital and sigma star one S. We would fill in the atomic orbital's and then distribute those electrons into our molecular orbital's here. So here we have for instance this could be the atomic orbital of hydrogen or helium and the other hydrogen or helium. Their valence electrons are found in the one S orbital for in period one of the periodic table. And remember we're looking at the valence electrons for these elements in hydrogen and helium. So hydrogen and di atomic helium which is not a normal structure. So we take a look here at molecular orbital diagrams in this first one. Now as a result of increase in electro negativity and a decrease in atomic size. Now recall that a whole nuclear diatonic molecule is composed of two identical elements bonded together.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
